At the end of the previous Single Scoop, Norm Horowitz, Jerry Hubbard, and George Hobby were ready to fly to Mars their experiment (the HHH experiment) to detect fixation of inorganic carbon (14CO2 and/or 14CO) in Martian soils to give 14C-labeled “higher organics”, when the soil was illuminated. This experiment was intended to detect Martian photosynthesis. This scoop describes what they observed, and how they actually interpreted their observations.
The TLDR Summary
• Viking soils converted large amounts of gaseous 14CO2 and/or 14CO into non-gaseous 14C-labeled material that was not flushed out of the soil at 120°C, but was released from the soil at 635°C.
• 99% of the released label was evidently in the form of 14CO2, as it did not absorb to Chromosorb P. This corresponded to ~100,000 Earth cell equivalents of fixed carbon.
• The remaining 1% of the released label was evidently in the form of 14C-labeled organic fragments, as it did absorb to Chromosorb P. This corresponded to ~1000 Earth-cell equivalents.
• The team interpreted the 1% fraction as 14CO2 fixed by photosynthetic Martian microbes into organic molecules, an affirmative sign of life.
• The team assigned the 99% fraction as 14CO2 that was absorbed onto the walls of the reactor or the Martian soil at 120 °C, but released at 635 °C, They did not mention its possible origin from 14C-label arising from 14CO2 fixed by Martian autotrophs at low temperature, then oxidized at 635 °C by nitrate in the Martian soils, even though they had previous seen exactly this in the Atacama Desert soils in Earth tests.
• One modern interpretation is that the Martian soil fixed enough 14CO2 into organics for ~100,000 cells, where 99% of the fixed organics were burned by perchlorate, discovered on Mars in 2009. The 1% corresponds to methyl chloride, dichoromethane, and other chlorinated organic fragments that are known to be the products of perchlorate oxidation of organics. Dichloromethane is also known to absorb on Chromosorb P.
• However, some fraction of the 99% might arise from 14CO2 bound as inorganic bicarbonate and carbonate, where these were not decomposed at 120 °C, but were decomposed at 635 °C.
• Some fraction of the 1% might be 14CO2 from inorganic bicarbonates and carbonates that absorbed in small amounts to Chromosorb P, despite multiple controls that suggested that this absorption did not happen.
Details for the Astrobiology Students.
To refresh your memory from the last scoop, the HHH experiment had extremely sound biological premises, then and now:
• All life requires higher organic molecules, molecules made from carbon atoms.
• 14CO2 and 14CO are the major sources of carbon in the Mars near-surface environment.
• Thus, if Martan life exists at the Viking sites, it must make 14C-labeled higher organics from 14CO2 and/or 14CO.
• This carbon fixation could be measured by measuring the amount of radioactive 14C in the Martian soil after exposing samples of that soil to 14CO2 and/or 14CO for five days.
• As a detail, should Martian microbial carbon fixation follow chemistry in any way analogous to carbon fixation by Terran photosynthetic microbes, some fixation would be observed for a time even in the absence of light.
• However, as a control, no fixation should be seen in samples that had been sterilized by heating the soil to 170 °C, after which no biological activity was presumed to be possible.
• Very few non-biological processes convert 14CO2 and/or 14CO into compounds that contain C-C bonds, and even fewer that contain C-H bonds.
The Carbon Assimilation Experiment as Done on Mars
The only challenge in the experimental design from with the difficulty of counting 14C-radioactivity in soils. The difficulty arises because the soil absorbs the high energy electrons (often called “beta particles”) emitted from 14C-atomic decay before they can (efficiently) reach a detector.
Accordingly, HHH experiment used a (somewhat) complex workflow to get the 14C-atoms in higher organic molecules out of the soil and into a radiation detector. In detail, the workflow had these steps (Figure 1):
Step 1. A scoop of Martian soil was delivered to an enclosed chamber with a light bulb, delivered from Earth to make sure it did not emit harsh UV light that might create the false positive discussed in the previous Scoop.
Step 2. A mixture of 14CO2 and 14CO gases brought from Earth was put into the chamber.
Step 3. The light was turned at temperatures that ranged from 8 °C to 26 °C.
Step 4. Any photosynthetic microbes in the soil were given 120 hours (5 days) to fix as much radioactive carbon as they could into higher organic molecules.
No water was added, avoiding an unnatural flood of water that might kill Martian microbes inexperienced to it. The experiment was entirely natural, except for the label, the exact illumination (about 20% of the intensity in the relevant wavelengths, the short wavelengths being cut off), a 28% higher CO2 atmospheric pressure (from 7.6 millibar to 9.8 millibar), and a ~23 fold higher partial pressure of CO. Then:
Step 5. The soil sample was heated to 120 °C to vent the excess radioactive 14CO2 and 14CO gas that had not been fixed, but possibly remaining adsorbed to the soil as 14CO2 and/or 14CO, not fixed into higher organic molecules by Martian microbes. This heating was seen on Earth to leave most biological higher organics intact.
Now the experiment was ready to go look for the 14C-radiolabeled higher organics in the soil. Again, the next steps in the workflow were intended to get radiolabel in those bio-organics out of the soil and into a spot where the soil would not interfere with its detection, the workflow continued:
Step 6. The soil sample was heated further, this time to 635 °C. This was intended to fragment, or pyrolyze, the fixed non-volatile 14C-labeled higher organics to give smaller organic fragments that were volatile. These were analogous to the small organics that come out of burnt popcorn to give the familiar burnt popcorn smell.
Step 7. The 14C-labeled organic fragments were then trapped in the pores and channels of the Chromosorb-P® brand of diatomaceous earth.
So now the 14C label from the fixed higher organics was safely out of the soil and bound as organic fragments to the Chromosorb-P®. So the problem now was to get those 14C-labeled organic fragments out of the Chromosorb-P® and into a detector.
For this purpose, the Chromosorb-P® was coated with copper oxide (CuO). At the low temperature where any 14C-labeled organic fragments arising by pyrolysis of the Mars-life-fixed organic were absorbed by the Chromosorb-P®, copper oxide did nothing. However, when Chromosorb-P® is heated to 640 °C, copper oxide oxidizes those 14C-labeled organic fragments to give radioactive 14CO2.
Now, the 14CO2 does not stick to Chromosorb-P®. Thus, the 14CO2 created from the 14C-labeled organic fragments would be blown out of the Chromosorb-P® and into a detector, where the energetic electrons released by 14C-radioactive decay would be counted, without interference from either the soil or the Chromosorb-P®. This was called Peak 1.
This gives the last three steps in the workflow.
Step 8. Heat the Chromosorb-P® with embedded copper oxide to 640 °C. This oxidized any trapped 14C-labeled organic fragments to 14CO2.
Step 9. Blow the resulting 14CO2 out of the Chromosorb-P® into a radioactivity detector.
Step 10. By measuring the number of beta particles emitted from the 14CO2 in the detector, quantify how much 14C had been fixed in the Martian soil by putative Martian microbes during the 5-day photosynthetic incubation.
A picture is worth a thousand words. So here it is.
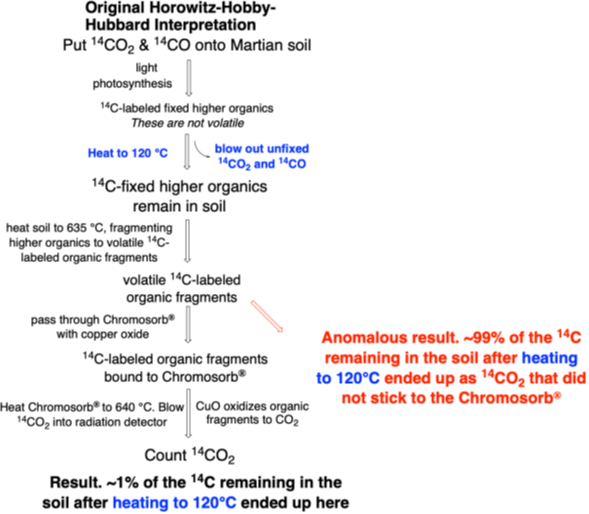
Figure 1. Workflow of the HHH carbon fixation experiment. As an anomaly, a surprisingly large amount (99%) of the total of the radioactivity remaining in the soil after the soil was heated at 120 °C, but released only at 635 °C. It did not bind Chromosorb-P® as expected for organic fragments, but passed through the Chromosorb-P® as if it were CO2 (Horowitz et al., 1977)
The Carbon Assimilation Results on Mars
So, what was actually seen? The HHH team reported that they saw a significant amount of radioactive 14CO2 released from the copper oxide-impregnated Chromosorb-P® upon high heating. This is Step 8 in the workflow. According to the experiment’s design, this 14CO2 could have come only from organic molecule fragments released pyrolytically from the Martian soil. Those fragments could be radiolabeled only if they had come from 14C-labeled higher organic molecules that the soil had fixed using the radiolabeled 14CO2 and/or 14CO gases that Viking had carried from Earth to Mars and presented to the soil in Step 2.
Under the terms of the experiment, Horowitz, Hobby, and Hubbard had discovered autotrophic Martian life.
Only about 1% of the 14C radioactivity remaining in the soil after it had been heated to 120 °C ended up sticking to the Chromosorb-P®. Only this 1% could reliably be interpreted under the terms of the experiment as arising from carbon fixation by Martian autotrophs. But these amounts were substantially more than what the team called “experimental error”. For those familiar with statistics, the team reported that this radioactivity was “three sigmas above background”. In other words, something in the Martian soil was converting significant amounts of 14CO2 and/or 14CO into 14C-labeled organic matter.
In other words, the HHH experiment found autotrophy on Mars. As we will discuss below, the amount of fixed 14C corresponded to the amount of carbon in ~1000 E. coli cells. And this conclusion survives fairly robustly even today.
Some 14C fixation was also observed when the experiment was repeated, but this time without light. But carbon fixation by photosynthetic autotrophs on Earth occurs also in the dark, at least until the point where they run out of energy and need another dose of sunlight. This process is appropriately called “the dark reaction”.*
Another observation surprised the Viking team. The soil continued to fix 14CO2 even after it had been heated to 90 °C. While the ability of Martian life to tolerate heating was not known, many Terran photosynthetic autotrophs die when heated at 90 °C. Including likely the plant on your windowsill. The putative Martian autotrophs seem to be especially tough.
In any case, stronger heating to 175 °C destroyed the ability of the Martian soil to fix 14C. That, the experimenters presumed, was hot enough to kill any microbe, Terran or Martian.
In short, under the terms of the experiment’s design, every detail of the results from the carbon fixation experiment was consistent with a Martian soil containing a photosynthetic autotroph. This Martian microbial “plant” appeared to be a bit more resistant to heat than we expect from our experience with Terran photosynthetic autotrophs. Otherwise, the results were the same. And later discoveries have not challenged this interpretation.
The HHH team left an anomaly without a robust interpretation
So far so good. But there was an anomaly. When the HHH team heated the Martian soil to 635 °C, they observed an unexpectedly large amount of radioactivity coming from the soil that did not stick to Chromobsorb-P®. This anomalous result is highlighted in red in Fig. 1.
And it was not a small amount of radioactivity. It was 100 times more than the radioactivity that did stick to Chromobsorb-P®, the blue step in Fig. 1. This radioactivity was interpreted to be 14CO2 coming from the soil.
But where did the 14CO2 come from? Why was it there? The Earth based experiment suggested two possibilities. First, this could have been 14CO2 absorbed to the alkaline soil as bicarbonates and carbonates. But the absorption must be sufficiently strong that the absorption would not have been reversed at 120 °C. It must not have been flushed out when the soil was heated to 120 °C in Step 5.
As a second possibility, this large amount of 14CO2 could represent carbon atoms from fixed organics that got their oxygen from other species in the soil. As we will see, the modern interpretation of the source of that oxygen is perchlorate (ClO4–) in the soil.
Remarking on this unexpectedly large amount of 14CO2 released in Step 6, Horowitz wrote (Horowitz et al., 1977), with my comments inserted in square brackets:
“The lamp is then turned off, and the chamber is brought to 120 °C while the radioactive atmosphere is vented [Step 5 in the workflow]. The chamber is next heated to 635 °C to pyrolyze organic matter in the sample [Step 6]. The volatile products, together with a large amount of 14CO2 and 14CO desorbed from the soil grains and walls of the chamber, are swept by a stream of He into a column packed with a mixture of 25% cupric oxide and 75% Chromosorb-P® [Step 7]. The column, which operates at 120 °C, retains organic molecules larger than methane but allows all but a small fraction of the CO2 and CO to pass into a radiation counter where their radioactivity is measured. This count is referred to as peak 1.”
The anomaly is in italics.
Now, the HHH team called the radioactivity in the unexpected 14CO2 and/or 14CO “Peak 1”. Peak 2 was the radioactivity in 14CO2 that was formed from organic fragments that stuck to Chromosorb-P®, and then produced by copper oxide oxidation. And thus 99% of the radioactivity was anomalous.
But the HHH team did not puzzle long over it. They simply stated that this material had been absorbed onto the “soil grains and walls of the chamber”.
So what do we make of this? It is hard to believe that the chamber walls absorbed 14CO2 and/or 14CO even at 120 °C. This would have been observed when the instrument was tested pre-flight on Earth. The team would never have flown a chamber that did this.
Of course, the pre-flight experiments on Earth did not have access to Martian soil. Thus, it remains a possibility that alkali in the Martian soil, but not in the Terran soils used to develop the instrument, made carbonates and/or bicarbonates that gave the feared false positive.
So here we have an anomaly. A large amount of 14CO2 and/or 14CO was allegedly sticking to the soil and chamber walls at 120 °C, and not flushed out in Step 5. And this is where we must close for the day. In the next Scoop, we will explain the how this anomaly was dismissed in real time, and how it might be interpreted today.
References
Horowitz, N. H., Hobby, G. L., & Hubbard, J. S. (1977). Viking on Mars: the carbon assimilation experiments. Journal of Geophysical Research, 82(28), 4659-4662
Posted with minor revisions by Jan Spacek 2/20/2026.
Spacek’s notes:
* Carbon fixation through atmospheric chemosynthesis is common in Earth’s cold deserts, at some locations the trace gas metabolism (not reliant on sunshine) is the main source of organic carbo in sparse cold soils. I personally believe that on Mars the photosynthesis is less likely than chemosynthesis – however, this was not known when Viking experiments were designed.