At the end of the previous single scoop Norm Horowitz, Jerry Hubbard, and George Hobby had collected on Mars data from the HHH experiment seeking to detect the fixation of inorganic carbon (14CO2 and/or 14CO) by Martian soils to give 14C-labeled “higher organics”. After five days, pyrolysis of the soil exposed to these radioactive gasses gave organic fragments that indicated fixation of enough carbon for about 1000 Earth-like cells. They interpreted these results in 1976 as evidence for autotrophic life on Mars. That interpretation has remained robust for a half century, despite much more information having come from Mars since.
However, results of a separate gas chromatography-mass specctrometry (GC-MS) experiment were (incorrectly) interpreted as having shown that the Martian soils contained no organic molecules. Gerald Soffen, the Viking Chief Scientist, declared: “That’s the ballgame. No organics, no life”. Further misinterpretations of other experiments led to a “consensus view” that the Martian soil contained highly reactive oxidizing agents (“peroxides” of unknown form represent the “consensus”) that accounted for the absence of meteoritic organics, generated O2 gas release upon soil humidification, and released 14CO2 from radiolaeled food. All of these “consensus” views have been known to be wrong since 2010. However, they persist even until 2026, in NASA-endorsed workshop documents, Decadal Surveys, DARES workshops, and authoritative publications from community members.
In a fascinating story of sociology, in 1977, Norm Horowitz himself discounted the results of the experiment that he reviewed. Even as he did, however, he noticed the contradictions in the consensus interpretation. As is often the case in science, those contradictions did not lead him to question the “consensus” and correct its mistakes. Rather, the contradictions were “explained away.” In an object lesson for students, this was done in part by assuming the outcomes of experiments that had not yet been done.
The “consensus” by 2005 was represented in textbooks in the form of statements that the HHH experiment failed to detect fixation of any organics. One taught, in direct contradiction of fact, that “no detection of [fixed] radioactive carbon was made, suggesting that no photosynthesis actually occurred during the experiment.”’” (Lunine, 2005). Today, the Horowitz Wikipedia page states that “Horowitz’s pyrolytic release experiments in 1976 provided the first indication that there is no current life on the surface of Mars”.
Here, we examine how “yes” became “no’ in the HHH experiment.
The TLDR Summary
• Viking soils were observed to convert large amounts of gaseous 14CO2 and/or 14CO into non-gaseous 14C-labeled material that was not flushed out of the soil at 120°C, but was released from the soil at 635°C.
• 99% of the released label was evidently in the form of 14CO2, as it did not absorb to Chromosorb P. This corresponded to ~100,000 Earth cell equivalents. This may represent fixed organic carbon that was converted to 14CO2 by perchlorate oxidation. This would indicate a substantial biosphere, perhaps 100,000 cells per gram of soil.
• The remaining released 14C-label was evidently in the form of 14C-labeled organic fragments, as it did absorb to Chromosorb P. This corresponded to ~1000 Earth-cell equivalents per gram of soil.
• The team initially interpreted the 1% fraction as 14CO2 fixed by photosynthetic, an affirmative sign of life.
• The team dismissed the 99% fraction as 14CO2 that was absorbed onto the walls of the reactor or the Martian soil, without mentioning its possible origin from 14C-label arising from 14CO2 fixed by Martian autotrophs at low temperature, then oxidized at 635 °C by nitrate in the Martian soils, even though they had previous seen exactly this in the Atacama Desert soils in Earth tests.
• One modern interpretation is that the Martian soil fixed enough 14CO2 into organics for ~100,000 cells per gram of soil, where 99% of the fixed organics were burned by perchlorate, and 1% emerged as methyl chloride, dichoromethane, and other chlorinated organic fragments that are, since 2010, known to be products of the oxidation of organics by perchlorate. Dichloromethane is known to absorb on Chromosorb P.
• However, some fraction of the 99% might arise from 14CO2 bound as inorganic bicarbonate and carbonate, where these were not decomposed at 120 °C, but were decomposed at 635 °C.
• Some fraction of the 1% might be 14CO2 that absorbed to Chromosorb P, despite multiple controls that suggested that this absorption did not happen. The HHH team went to lengths to exclude this artifact, however.
• However, after Gerald Soffen had “called the ballgame”, perhaps to conform to the community “consensus”, Horowitz abandoned his life-affirming interpretation. He accepted the “consensus” that the Martian soil contained a highly reactive oxidant.
• Nevertheless, Horowitz wrote this:
“[I]t was surprising that in such a strongly oxidizing environment even a small amount of organic material could be fixed in the soil. It is not easy to point to a nonbiological explanation for this result”.
• Nevertheless, Horowitz went with the “consensus” writing:
“Investigations into the problem are now under way in terrestrial laboratories with synthetic Martian soils formulated on the basis of the data from the inorganic analyses carried out by the Viking landers. A solution to the puzzle will probably also explain why the organic-analysis experiment detected no organic material in the Martian surface.”
• As the lesson to the strobiology student, never rely on assumed outcomes of experiments that have not yet been done.
Details for the Astrobiology Students
Where we left the story last, Norm Horowitz, Jerry Hubbard, and George Hobby had run the “HHH” experiment on Mars, delivered there at two sites by Viking 1976. They saw the conversion of gaseous inorganic carbon (14CO2 and/or 14CO) by Martian soils into non-gaseous in the form of 14C-labeled “higher” organics. About 1% of this was released from the soil in the form of materials that absorbed on Chromosorb. Under the terms of the experimental design, these were fragments of organics fixed in the soil by Martian autotrophs. The remaining 99% of the label came off the soil in the form of 14CO2.
In preflight experiments on Earth, the HHH team had noted that if the soil was alkaline, then 14CO2 could be absorbed into the soil as bicarbonate or carbonate salts. Also in pre-flight experiments, the HHH team had noted that if the soil contained oxidants like nitrate, fixed organics could be burned to give 14CO2.
These observations gained a new possible interpretation in 2009. In that year, the Phoenix Lander in the Martian Arctic found perchlorate (ClO4–) in Martian soils (Hecht et al., 2009). And perchlorate was not present in small amounts. It was 0.4 to 0.6 percent by weight of the soil. Perchlorate was also present in some samples of Atacama soil, but only 0.03 percent by weight. In the Atacama, the dominant oxidizing salt is nitrate.
Now, perchlorate is not nitrate. However, like nitrate, perchlorate does nothing to higher organics at low temperatures. However, when heated with organics, perchlorate also sets them on fire. The principal product is CO2, with a percent or two of CH3Cl and CH2Cl2, depending on the precise structure of the organics being oxidized. CH2Cl2 is known to absorb on Chromosorb-P®. CH3Cl is not known to absorb on Chromosorb-P®, but I expect that it does. Later work also saw CHCl3, CCl4, and other chlorinated organics.
In 1976, the HHH team did not mention nitrate-mediated oxidation of higher organics as a source of the 14CO2 that passed through the Chromosorb-P®. Here is how they report their observations on the surface of Mars (Horowitz et al., 1977):
“The lamp is then turned off, and the chamber is brought to 120 °C while the radioactive atmosphere is vented [Step 5 in the workflow]. The chamber is next heated to 635 °C to pyrolyze organic matter in the sample [Step 6]. The volatile products, together with a large amount of 14CO2 and 14CO desorbed from the soil grains and walls of the chamber, are swept by a stream of He into a column packed with a mixture of 25% cupric oxide and 75% Chromosorb-P [Step 7]. The column, which operates at 120 °C, retains organic molecules larger than methane but allows all but a small fraction of the CO2 and CO to pass into a radiation counter where their radioactivity is measured. This count is referred to as peak 1.”
It is curious that, in 1977, Horowitz did not mention either the possibility of 14CO2 being absorbed onto alkaline Martian soils or the possibility that the 14CO2 could arise from nitrate oxidation of fixed organics. Thus, the dismissal of the 14CO2 in anomalously large amounts would be dismissed. However, Horowitz continued:
“The [Chromosorb-P] column temperature is then raised to 640 °C [Step 8], the high temperature causing the release of organic compounds and their oxidation to CO2 by the CuO in the column. The radioactivity of this gas is peak 2: it represents organic matter synthesized from 14CO or 14CO2 during the [photosynthetic] incubation. Peak 2 also contains the small fraction of CO and 14CO2 which failed to elute with peak 1, presumably because of the presence of some high-affinity sites in the [Chromosorb-P] column. The radioactivity of this fraction, referred to as peak 2(0), must be subtracted from peak 2 in order to estimate the amount of C fixed in organic matter.”
The HHH team evidently did not consider the possibility that both Peak 1 and Peak 2 radioactivity had come from bio-fixed bio-organics, but combusted by nitrate in the soils. Even though their work on Earth prior to the Viking mission had seen exactly that. They evidently did not consider the possibility that the Martian soils contained more nitrate than the Atacama soils.
As we shall see in the next sets of single scoops when we discuss gas chromatography-mass spectrometry results, separate experiment that heated the Martian soil without adding any 14CO2 or 14CO saw the evolution of CO2, CH3Cl, and CH2Cl2. That is, it detected the perchlorate oxidation of organics in the Martian soil. Thus, since 2009, the HHH experiment could have the interpretation shown in Fig. 1.
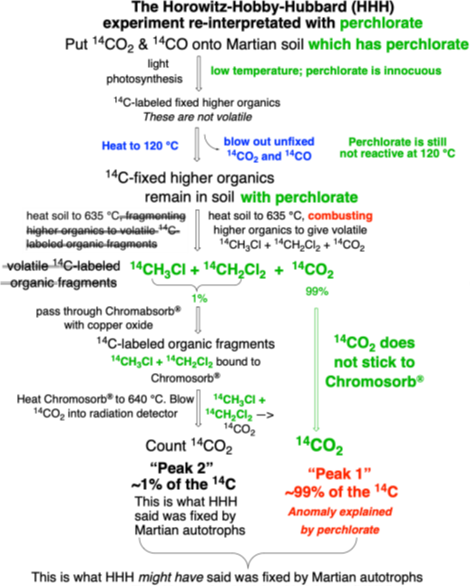
Figure 1. With perchlorate in the Viking soils, an alternative interpretation is possible for the HHH experimental results. Here, the analysis recognizes that excess perchlorate burns higher organics to give 14CO2 (which does not stick to Chromosorb-P®) and CH3Cl and CH2Cl2 (the last is known to stick). Thus, the amount of 14C fixed by Martian soil autotrophs might be represented by the sum of radioactivity in Peak 1 and Peak 2. This is 100 times more than what the HHH team reported by way of fixed organics, and corresponds to ~100,000 Earth-like cells per gram of soil.
Figure 1 shows a re-do of the Figure 1 in the previous Single Scoop considering the presence of perchlorate in the soil. We can now go back to the numbers. If Peak 2 is seen to be the only material representing fixed organic molecules, the HHH experiment found that ~ 30 picomoles of carbon is fixed, that is 3 x 10-11 moles of carbon. This is a back of the envelope calculation, not including the fact that the 14CO2 was diluted 4:1 from atmospheric 12CO2. As I noted in an earlier scoop, an E. coli cell has 1 x 10-14 moles of carbon. So if the observations were interpreted as the HHH team did in 1976, the Martian soil fixed enough carbon for ~1000 cells. That is quite a bit, similar to the number of microbes in hyper-arid soils on Earth.
But if the radioactivity in Peak 2 is also seen as arising from fixed organics, here by perchlorate combustion, then the Martian soil samples fixed enough carbon for ~100,000 cells.
That is quite a biosphere.
But you see the problem. In the run-up to the Viking launch, the HHH team had identified the potential for a false positive, the non-biological absorption of 14CO2 as bicarbonates and carbonates with sufficient stabilities to not be flushed out at 120 °C. Perhaps their thought was to see Peak 1 as the result of this non-biological carbonate background. In any case, that is how they interpreted Peak 1 in 1976 on Mars.
But this runs ignores the false negative problem that the HHH team had clearly understood in their pre-flight work. In 1970, the HHH team understood that if nitrate were present in the Martian soils, as it was in the Atacama soils, some of the Peak 1 radioactivity could also come from fixed organics that were burned by nitrate at 635 °C. Indeed, if nitrate were in excess over organics, then all of the Peak 1 radioactivity might represent autotrophic fixation.
Evidently, no one considered the possibility of large amounts of nitrate in the soil that would not fragment the higher organics, but would convert them into 14CO2. The community “consensus” still does not recognize that with perchlorate, giving CH3Cl and CH2Cl2 as minor products, these couldhave been the pyrolysis products absorbed on Chromosorb-P®.
Of course, the false negative could be managed by counting the radioactivity in Peak 1 as part of biological carbon fixation. But not if the false positive problem is lurking.
Carbon was Evidently Fixed on Mars
This allows us to consider interpretations at the extremes of possibility space. If nitrate, perchlorate, and all other oxidants were entirely absent from the Martian soil, the HHH 1976 interpretation would be correct. The only radioactivity representing carbon fixation was found in Peak 1, enough for ~1000 Earth-like cells. All of Peak 2 was thus 14CO2 that was non-biologically fixed on alkaline soils as bicarbonate or carbonate salts, and not flushed out at 120 °C. These bicarbonates/carbonates released their radioactivity at 635 °C, according to this interpretation.
At the other extreme, if perchlorate or another oxidant were present in excess over fixed organics, then Peak 2 represents chlorinated combustion products arising from the reaction of higher organics with perchlorate. These can be directly attributed to higher organics, as they were in the HHH 1976 interpretation, although with an interpretation different from the 1976 HHH interpretation.
However, we cannot a priori determine the fraction of the radioactivity in Peak 2 to attribute to perchlorate combustion of higher organics, and which to attribute to non-biological absorption of 14CO2. Now, if we knew the ratio of CO2:CH3Cl:CH2Cl2 products from bio-organic molecules burned by perchlorate in general, we might estimate the portions of Peak 1 14CO2 that came from fixed bio-organics from bicarbonate/carbonate inorganics. But we do not know this ratio.
This sets up the next Single Scoop, to ask: What experiments are needed now on Earth to understand the extent of carbon fixation in Martian soils, what fraction of that are to be ascribed to biological processes, and what fraction should be ascribed to non-biological processes. This will lead to the same questions for future Mars missions.
References
Hecht MH, Kounaves SP, Quinn RC, et al. Detection of perchlorate and the soluble chemistry of martian soil at the Phoenix lander site. Science 2009; 325(5936):64-67; doi: 10.1126/
science.1172466
Horowitz, N. H., Hobby, G. L., & Hubbard, J. S. (1977). Viking on Mars: the carbon assimilation experiments. Journal of Geophysical Research, 82(28), 4659-4662
Posted with minor revisions by Jan Spacek 2/20/2026.