Reviewing the story, the HHH experiment to seek carbon fixation in the near-surface of Mars was completed in 1976. It had seen enough of this autotrophy to support the formation of ~1000 Earth like cells, three sigmas above background. The observable was 4C-radiolabel emitted from soil heated to 635 °C and captured by Chromosorb-P®, organic fragments that could be labeled only if the soil had an activity that reduced 14CO2 to make molecules with C-C bonds. Although the amount was small, it was consistent with bio-loads observed in resource starved soils on Earth. Accordingly, in 1976, this was seen as an affirmative sign of life.
However, there were these problems:
(a) The signal that (nearly) indisputably could be attributed to this activity was weak, just three sigmas above background, Yes, that corresponded to the number of cells expected in this particular resource poor environment if we use Earth analogs, ~1000 Earth-type cells per gram. However, one generally worries about small signals.
(b) That signal was surprisingly robust to heating. Even though it did not survive at 175 °C, it did survive at 90°C. And unlike organisms in the Mojave Desert, which survive at 150 °C, no selection pressure on Mars makes obvious the fitness value of that level of thermostability.
(c) The signal remained at a lower level without illumination. This is entirely consistent with photosynthetic autotrophy, where fixation proceeds in a “dark reaction”. Alternatively, as commented by Jan Spacek in a previous Scoop, it is also consistent with non-photosynthetic autotrophy, for example, using H2 or CO as energy sources, something common in Terran microbiology in resource-sparse environments.
(d) A mechanism to manage a known potential for a false negative had not been managed, the possibility that nitrates converted 14C-14C organics into 14CO2. This would move “Peak 2” 14C-label from organic fragments trapped on Chromosorb-P and thus (essentially) indisputably arising from fixed organics, into “Peak 1” 14C-label. On Mars, 100 fold more radioactivity was observed in this Peak 1 14CO2 radioactivity than in radioactivity whose origin was assigned to fixed 14C-14C organics. If all of this Peak 1 radioactivity were combined with the Peak 2 radioactivity, then the HHH experiment saw the fixation of carbon sufficient for ~100,000 Earth-type cells.
(e) However, one cannot confidently make this combination, because of a false positive possibility that also was recognized, but not mitigated, by the HHH team as they prepared their experiment to fly. If the Martian soils were alkaline, some of the 14CO2 would be absorbed on the soils not by its transformation into organics, but rather as carbonates and bicarbonates. This label might have been flushed out at 120 °C, but it might not have been, but instead released only at 635 °C. It is possible that some of the Peak 1 label could contaminate the Peak 2 label, although the HHH team went to some lengths to rule this out.
(f) The 2009 discovery of perchlorate in Martian soils changed the story, due to the analysis by Rafael Navarro-Gonzales of its ability to convert organics to CO2, CH3Cl, CH2Cl2, and other chlorinated organics. Accordingly, the modern interpretation of the HHH experiment is that 14C-label was fixed into perchlorate-laden soils, remained there at 120 °C (where perchlorate is not an active oxidant), and then converted into 14CO2, 14CH3Cl, 14CH2Cl2, and other 14C-labeled chlorinated organics by perchlorate-mediated combustion at 635 °C. The 14CO2 went into Peak1, and the 14CH3Cl, 14CH2Cl2, and other 14C-labeled chlorinated organics went (by way of Chromosorb-P) into Peak 2. However, because the Peak 1 label could also arise from carbonates and bicarbonates stable at 120 °C but not at 635 °C, we cannot, without further analysis, estimate the actual amount of autotrophy seen on Mars by the HHH experiment.
(g) Further, the conversion of 14CO into 14C-14C organics by photochemistry, remains in the community a possibility for generating false positives. This process is well known, but requires short wave UV light { DOI:10.1007/BF00931407}. The HHH team had recognized this possibility in the planning phase, and had mitigated it by transporting to Mars a light filtered to exclude light with wavelengths M ~ 320 nm.
(h) However, after the “community” arrived at the “consensus” that life was not possible in “strongly oxidizing” Martian soils, the HHH team (evidently) was driven to find non-biological explanations for results that were life-affirming, under the terms of the experimental design. Thus, later in the decade, Jerry Hubbard attempted to observe photochemical fixation at longer wavelengths experimentally. The experiments “failed to reproduce Viking results”. Indeed, they could not reproduce the “dark reaction”. Nevertheless, those experiments have entered the “culture” as an acceptable non-biological explanation for the three sigmas of 14C-label fixation.
A hierarchy of challenges for astrobiology students
So now what? It is less important to teach students the “facts” about Mars (which, as we have seen, can change as they are driven by culture) than it is to teach you how to think about those facts. Accordingly, I lay out a set of challenges, together with one of several possible solutions.
In the first set, we go back in time to the early 1970s. Knowing nothing more than what Horowitz, Hobby, and Hubbard knew then, how would you have designed the HHH experiment better?
Here, we begin with the problems that the HHH team identified, but did not manage. Recapitulating, these included
• One false positive problem, the possibility of label being absorbed as (bi)carbonates, that are not flushed out of the soil at 120 °C, and release 14CO2 at 635 °C. While the HHH team sought to be certain that none of this Peak 1 signal contaminated the Peak 2 signal, this remains a possibility, especially if the signal is small. Further, if some of the Peak 1 signal arises from nitrate or perchlorate oxidation of fixed organics, the problem with (bi)carbonates could cause an underestimate of the amount of fixed organic.
• One false negative problem, arising from the oxidation of fixed organics by nitrate or perchlorate to convert Peak 2 label into Peak 1 label.
How would you have modified the workflow to manage the false positive problem better than the HHH team did?
To refresh your memory, here was the workflow annotated to highlight current interpretations.
Step 1. A scoop of Martian soil was delivered to an enclosed chamber with a light bulb, filtered to remove harsh UV light that might create the false positive from carbon monoxide.
Step 2. A mixture of 14CO2 and 14CO gases brought from Earth was put into the chamber.
Step 3. The light was turned on or, in a control, not turned on.
Step 4. Any photosynthetic microbes in the soil were given 120 hours (5 days) to fix as much radioactive carbon as they could into higher organic molecules.
Step 5. The sample was heated to 120 °C to vent the excess radioactive 14CO2 and 14CO gas that had not been fixed, but possibly remaining adhered to the soil as 14CO2 and/or 14CO. This step does not clearly manage the false positive problem, as (bi)carbonates formed in alkaline soils need not have been flushed out at those temperatures.
Step 6. The soil sample was heated further, this time to 635 °C. This was intended to fragment, or pyrolyze, the fixed non-volatile 14C-labeled higher organics to give smaller organic fragments that were volatile. If the soil contained nitrate, perchlorate, or another source of oxygen, the organics would be combusted, and leave the soil as 14CO2 (the major product, which passes through the Chromosorb-P and is counted as Peak 1) and (with perchlorate) 14CH3Cl and 14CH2Cl2 (minor products, the second known to stick to Chromosorb-P).
Step 7. The 14C-labeled organic fragments were then trapped in the pores and channels of Chromosorb-P® with copper oxide.
Step 8. Heat the Chromosorb® with copper oxide to 640 °C. This converted trapped 14C-labeled organic fragments to 14CO2.
Step 9. Blow the resulting 14CO2 out of the Chromosorb-P® into a radioactivity detector.
Step 10. By measuring the number of beta particles emitted from the 14CO2 in the detector, quantitate how much 14C had been fixed in the Martian soil by putative Martian microbes during the 5-day photosynthetic incubation. This is “Peak 2” radioactivity.
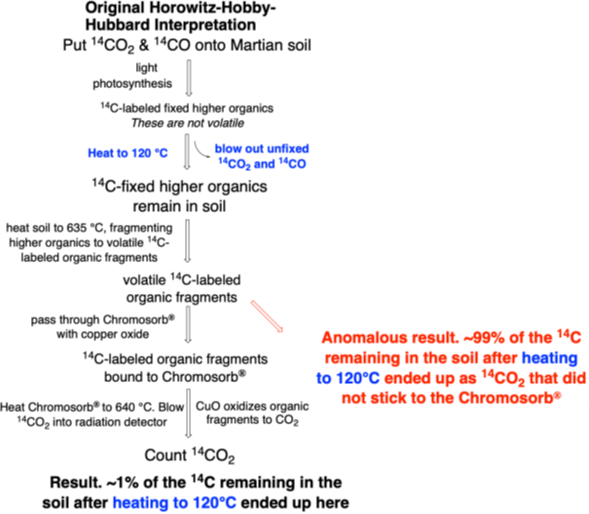
Figure 1. Workflow of the Horowitz carbon fixation experiment (Horowitz et al., 1977).
First, how could the HHH team have better managed the possibility of (bi)carbonate absorption on alkaline soils delivering Peak 1 radioactivity, which they realized would have made it impossible to assess the fraction of Peak 1 radioactivity that came from oxidative combustion of fixed organics?
Several strategies are possible, but here is one. Rather than flushing out the CO2 in Step 5 in a flow of gas, they could have added a volatile acid, such as hydrogen chloride, to the gas stream. The HCl would have destroyed the alkalinity of the soil and released any 14CO2 absorbed as (bi)carbonates. Then, pyrolysis can follow confident that (bi)carbonates are not the sources of the Peak 1 14CO2. This allows the Peak 2 organics to be more confidently attributed to the total amount of organic carbon biologically fixed, especially if nitrate- or perchlorate-effected combustion is a possibility.
This workflow modification can, of course, be tested on Earth, including with Atacama Desert soils. However, the modified workflow can also be incorporated as an experiment in an IMPRESS penetrator. Here, should the ride-share architecture be funded for development, it would be a low cost way to assess the biosphere on Mars at multiple sites where multiple penetrators might penetrate.
We can now move forward in time to ask how we might design an experiment that we can do on Earth to better interpret the data recovered in 1976 on Mars. The first experiment will fill in a gap in the literature. Thus, it is reported that Chromosorb-P captures 14CH2Cl2, but it is not reported that it captures CH3Cl. This experiment can be done by (i) obtaining Chromosorb-P from a supplier, (ii) obtaining CH3Cl in a lecture bottle, (iii) passing the CH3Cl through the Chromosorb-P, and (iv) measuring the weight gain. This will provide a “absorbing capacity” for the Chromosorb-P.
Then, to fill in the remaining gaps in the literature, the experiments would be repeated with copper oxide in the Chromosorb-P. This will complete the workflow from Steps 6-9. The process can then be reproduced with 14C-labeled organics in soils with and without oxidants.
Now, the ratios of CO2, CH3Cl, CH2Cl2, and other chlorinated organics depends on the structures of the organic molecules being oxidized. This is the case with other generic oxidation systems (e.g. the Kuhn-Roth oxidation). Indeed, in some cases, the oxidation patterns are so regular that analyses of the oxidation products was part of classical structure proof in organic chemistry. For example, the Kuhn-Roth oxidation process was used to determine the ratio of methyl carbon and non-methyl carbon in isolated steroids in classical organic chemistry.
Interesting, only the Sephton laboratory has attempted to connect the ratio of chlorinated products from perchlorate oxidation to molecular structures, but these only with complex oils. A set of experiments with defined organic molecules would allow us to constrain in a general way the structures of the organic molecules that were generated on Mars in 1976.
Further, if one is concerned about the possibility that 14CO is converted non-biologically into organics by long wave UV light that might be the source of false positives, these materials can be prepared in a Terran laboratory and oxidized by perchlorate to measure the ratio of CO2 and chlorinated organics.
Finally, to complete experiments that might be done on Earth, we can challenge the student to analyze thus prevailing wisdom for a non-biologic mechanism. Here, the question asks whether it (i) requires soil minerals to occur and, if it does, (ii) whether heating those soils to 175 °C destroys the process.
How might an IMPRESS architecture work?
Of course, the entire reason we have the problem is because the beta particles from 14C atoms are too weak to make it out of the soil. This is the reason for the workflow that converts an extremely simple concept (the conversion of volatile radioactivity to non-volatile radioactivity) into a Rube Goldberg work flow with pyrolysis, the complicating combustion, and fractionation of products on Chromosorb-P.
How would you re-design the experiment to detect the radioactivity directly in the soil?
In the laboratory, 14C is generally counted by placing the material in a hydrophobic solvent system based on toluene. This contains organic molecules known as scintillants, and also contains detergents. These generate ~300 photons for each beta particle that the isotope emits. If the scintillation fluid is placed on a CMOS surface that detects light, the label can be counted directly.
Here the workflow involves:
Step 1. A scoop of Martian soil is placed on top of a CMOS chip in an enclosed chamber with a filtered light bulb.
Step 2. A mixture of 14CO2 and 14CO gases is put into the chamber.
Step 3. The light is turned on or, in a control, not turned on.
Step 4. Any photosynthetic microbes in the soil are given 120 hours to fix as much radioactive carbon as they can into higher organic molecules.
Step 5. The sample is flushed in a stream of CO2 containing HCl to remove unfixed 14CO2 and 14CO gases as well as any (bi)carbonates formed in alkaline soils.
Step 6. Scintillation solution is added to the soil, percolates through the soil, and delivers organics to the surface of the CMOS chip
Step 7. The CMOS chip counts the radioactivity.
This workflow is also much less demanding from the perspective of instrumentation than the HHH experiment. In particular, it can be adapted for IMPRESS penetrator delivery.
References
Horowitz, N. H. 1977. “The Search for Life on Mars.” Scientific American 237 (5): 52–61. https://doi.org/10.1038/scientificamerican1177-52.